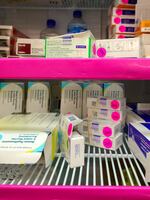
A refrigerator at Mid-Valley Children's Clinic in Albany, Ore., with supplies of government-funded vaccines for children on Medicaid. The pink "VFC" stickers help clinics follow stringent federal rules and ensure only eligible children get the "Vaccines for Children" shots.
Amelia Templeton/OPB
The Oregon Health Authority is expecting shipments of about 1,600 additional doses of the RSV shot Beyfortus for newborns by the end of the year, amid an ongoing shortage of the new therapy.
That’s unlikely to meet the current demand, according to OHA.
About 40,000 babies are born in Oregon each year, and federal health officials recommend all of them receive protection against the respiratory virus.
“We’re not going to have enough to give to all the babies that might qualify for nirsevimab, unfortunately,” said Dr. Paul Ceislak, the health authority’s medical director for immunizations.
The shot is recommended during RSV season for infants up to 8 months old whose mothers didn’t get the new RSV vaccine for pregnant people.
Nirsevimab comes in two different sizes: a 50 milligram dose for newborns, and a 100 mg dose for babies 11 pounds and up, who are typically around 2 months or older.
The vials for older infants have been in particularly short supply.
In a briefing for the American Academy of Pediatrics, a representative with the drugmaker Sanofi said the demand for the 100 mg doses exceeded the company’s supply for the entire season just weeks after shipping began.
Last month, the CDC issued an advisory asking clinicians to prioritize the larger doses for babies 6 months and under, and those 6-8 months with underlying health conditions that increase their risk for severe RSV.
FILE: A child receives a shot at vaccine clinic held at Clackamas Town Center, Nov. 10, 2021 in Happy Valley, Ore.
Kristyna Wentz-Graff / OPB
The new doses coming to Oregon are the smaller, 50 mg newborn dose. The state has already exceeded its maximum allocation of the particularly hard to find 100 mg doses for older infants, an OHA spokesman said.
Oregon isn’t issuing any of its own guidelines or rationing recommendations, because the state’s priority is making sure doses don’t go unused.
“If you have a baby who qualifies for Nirsevimab, and you have the product, administer it,” he said.
The shortage is challenging for families and providers as RSV season in Oregon is officially underway.
The percentage positive lab tests for RSV started rising in November. Cieslak expects cases to increase through the holidays and then taper off in the spring.
State immunization records show at least 4,306 infants in Oregon have received a dose of Nirsevimab so far this year. But that number is likely an undercount. In some cases, pediatricians aren’t required to report doses they’ve purchased and given to patients with commercial insurance.
The state expects to receive its additional newborn doses through Vaccines for Children, a federal safety net program that distributes free shots to providers for kids on Medicaid, uninsured children, and Alaska Native and American Indian children.
About half of all Oregon children qualify for free VFC shots.
Through the program, OHA will prioritize getting additional doses of Nirsevimab to federally-qualified health centers, tribes and local public health authorities.
The CDC has said that distributing Nirsevimab to Alaska Native and American Indian children should be a particular priority. In studies, those children had a four to 10 times higher risk of severe RSV than the general population.
State and federal rules bar providers from using doses of VFC shots for patients with commercial insurance.
OHA had little information about how families with commercial insurance have been impacted by the ongoing shortage, or whether health systems expect additional private market shipments.
Sanofi, the drugmaker, refused to answer questions about how many doses it has shipped to private market customers to date.
Kaiser Permenente Northwest said it has been able to order additional 50 mg vials from Sanofi for the newborn population, and likely has enough to cover newborn infants for the remainder of the RSV season.
However, Kaiser has a limited supply of the 100 mg doses and may run out in the next two months.
OHSU is also currently providing Nirsevimab at birth to all infants born in its hospitals, until its supply of 50 mg doses runs out.
Salem Health, the only hospital in the state participating in the VFC program, is offering the shot to all newborns at birth.
Many other hospitals have been unable to provide the shot to newborns at birth, due to how difficult it is to enroll in the VFC program and the high cost of the shot.
Families who can’t get access to the shot can follow longstanding guidelines to protect newborns and very young infants who are at the highest risk. That includes having family members who are sick wear masks or stay away from a new baby if possible. Adults should also wash their hands regularly and clean frequently touched surfaces.
For people who are currently pregnant, Pfizer’s maternal RSV vaccine, brand name Abrysvo, is approved as a way of protecting their babies against severe RSV after birth.
